Biomere’s scientists work across multiple therapeutic areas, including CNS diseases, oncology, and metabolic diseases. Our team has developed deep expertise in dosing routes for both large and small animal models, as well as extensive experience with complex rodent models for oncology and metabolic disease studies.
Home – Therapeutic Areas
A critical consideration in the preclinical evaluation of CNS-targeted therapies is the choice of dosing route. Systemic dosing often results in poor CNS distribution due to the blood-brain barrier (BBB), whereas tissue-specific dosing can significantly improve bioavailability in CNS tissues and cerebrospinal fluid (CSF).
The most commonly used CNS dosing routes in preclinical animal models, including rodents, rabbits, and non-human primates (NHPs), are:
Once a therapeutic is administered directly into CNS tissues, CSF sampling and analysis is the standard method for assessing drug exposure and biomarker levels. Serial CSF collection is facilitated by the placement of chronic indwelling ports, which enable repeated sampling in conscious animals and eliminate the need for multiple sedation events. Properly placed CSF ports also minimize sample contamination with red blood cells. Beyond chronic porting, Biomere’s expertise extends to direct-stick techniques targeting the lumbar and cisternal intrathecal space, for both IT dosing and CSF collection.
Biomere’s small animal scientists have deep expertise in rodent tumor models for evaluating novel antitumor therapies, including CAR-T, ADCs, monoclonal antibodies (mAbs), small molecules, and gene therapies. Once a tumor model is established, our team can perform a comprehensive suite of assays, including:
The team has experience with CDX (cell-derived xenograft), syngeneic, and humanized mouse models (e.g., NSG) to support evaluation of complex antitumor therapies.
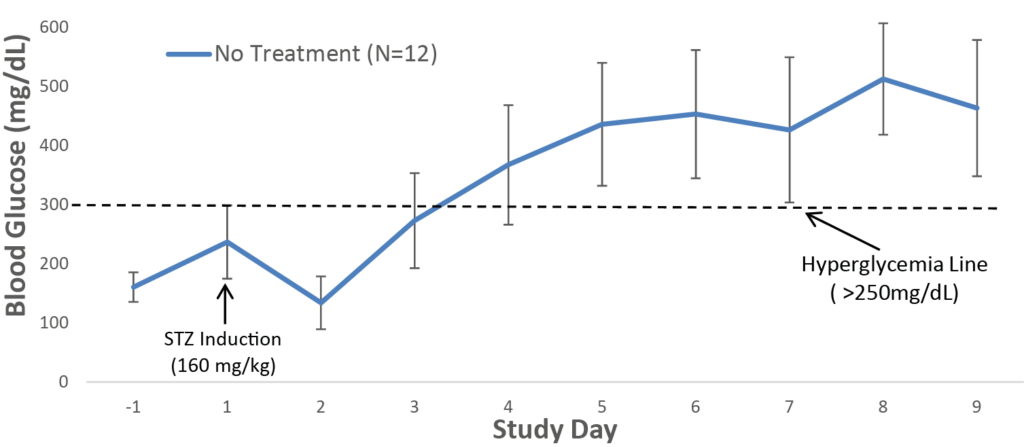
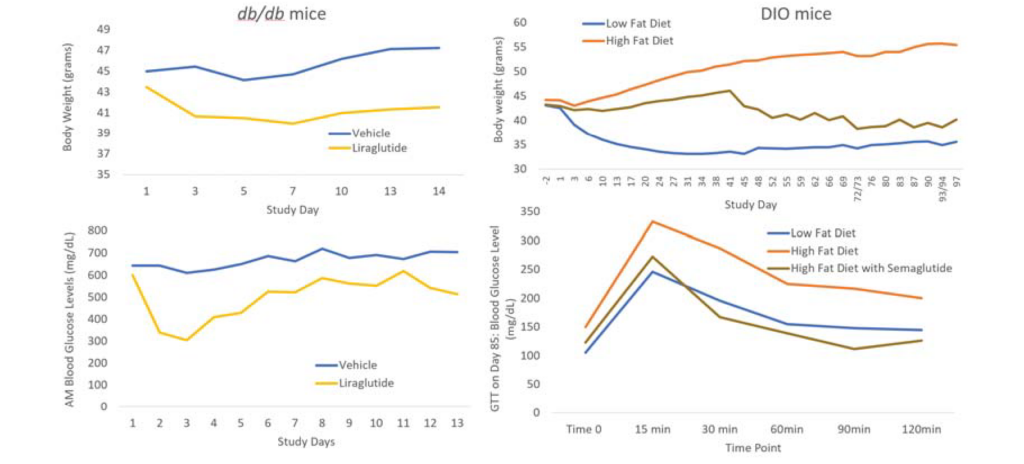
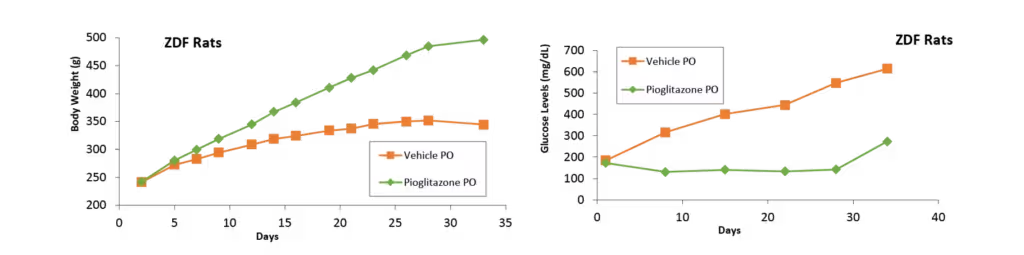
Biomere has experience with a broad range of rodent models for metabolic diseases, including obesity, type I and type II diabetes, hyperlipidemia, and hypercholesterolemia. Our team works with both induced and genetically modified models, including:
Relevant endpoints for metabolic disease models include body weight, blood glucose, insulin, and hemoglobin A1c (HbA1c). Biomere also offers metabolic cages for urine and fecal collection to support downstream analysis. Additional endpoints encompass cytokine levels and clinical chemistry markers, including HDL, LDL, total cholesterol, triglycerides, and liver enzymes. In partnership with specialist providers, Biomere offers comprehensive histology services including immunohistochemistry (IHC) and singleplex and multiplex immunofluorescence (IF).